A major step forward in making food, fuels, and medicines more efficiently and affordably has just been made, thanks to new research involving scientists from Oregon State University and several institutions in China.
The breakthrough centers on improving a key chemical process called hydrogenation, which is used in everything from food production to fuel refining.
Hydrogenation is a process where hydrogen molecules are added to other compounds, helping turn raw materials into useful products.
For example, it’s used to make margarine from vegetable oils and to produce cleaner fuels like propane and butane from crude oil.
But hydrogenation reactions need a catalyst—something that speeds up the reaction without being used up itself. Usually, expensive metals like platinum or palladium are used as catalysts.
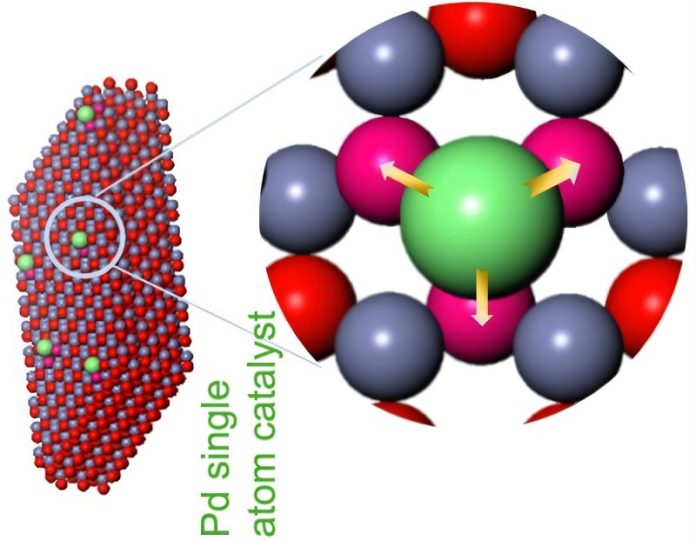
In this new study, published in the journal Nature, researchers focused on a cutting-edge type of catalyst known as a single-atom catalyst (SAC).
Unlike traditional catalysts made of clusters or nanoparticles, SACs are made of individual metal atoms spread out on a supporting surface.
These tiny, isolated atoms can be more efficient and cost-effective—but scientists didn’t fully understand why they worked so well.
Led by Zhenxing Feng, an associate professor at Oregon State University, and in collaboration with scientists in China, the team created 34 versions of palladium single-atom catalysts placed on 14 different supporting materials made of semiconductors. They studied how well these SACs performed using advanced tools like X-ray and infrared imaging.
The researchers discovered that the performance of these catalysts depends on how well the supporting material can accept electrons.
In simpler terms, if the material holding the metal atom is good at pulling in electrons, the reaction works better. Even better, they found this relationship follows a clear pattern—meaning scientists can now predict which combinations of metals and support materials will work best.
They also found that making the support material smaller improved the catalyst’s power even more.
The study focused on a common industrial reaction: turning acetylene into ethylene, which is a key ingredient in many plastic products. Thanks to this discovery, industries could soon create products more efficiently while using fewer expensive materials.
This breakthrough could help make hydrogenation cheaper, cleaner, and more sustainable—benefiting everything from food production to energy and the environment.
Source: Oregon State University.



