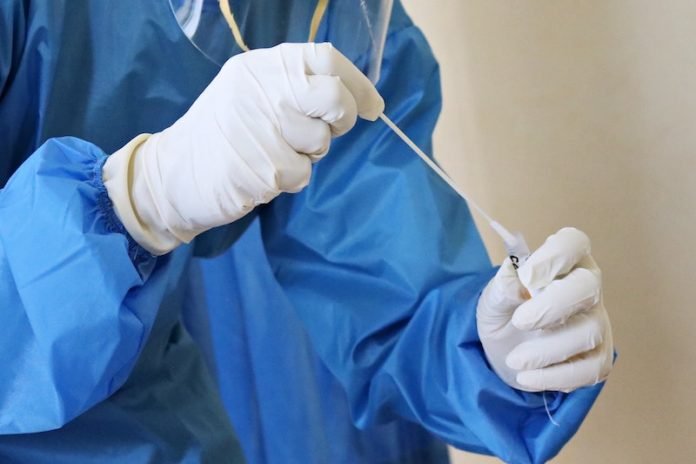
Until a vaccine is available, curbing the coronavirus pandemic relies heavily on how quickly a potentially exposed individual can be tested and quarantined.
However, the current diagnostic techniques vary in reliability and relevance, so an understanding of which test is most appropriate for a given circumstance is necessary to avoid false reports.
In a new study, researchers tested the available diagnostic techniques and determined key steps required for better testing moving forward.
The research was conducted by a team from Van Yuzuncu Yil University and elsewhere.
Laboratory tests that target the virus’s genes—known as real-time reverse transcription-polymerase chain reaction assays—are currently the gold standard for testing. But according to the Food and Drug Administration, these could give false negatives.
These tests depend on the presence of antibodies, which may not have yet been developed in the early stages of infection.
Since different antibodies appear at different stages, diagnostic tests must be chosen to target the appropriate immune response based on when an individual is believed to have been infected.
The team emphasizes point-of-care testing as an urgent objective.
These types of tests would help detect the virus on-site without the need for laboratory equipment or specialized personnel, eliminating or reducing the wait time between testing and obtaining results.
A quite sensitive test that can measure the existent tiny number of viral particles, or any parameter related to the particle—weight, structure, charge, diameter—can provide rapid and early diagnosis.
When asked about the potential for a more comfortable testing method, the team stressed that this depends on the sampling method and its sensitivity.
A fingertip blood test or a saliva test could potentially be underway, though these have their own drawbacks.
One author of the study is Oguzhan Gunduz.
The study is published in the journal APL Bioengineering.
Copyright © 2020 Knowridge Science Report. All rights reserved.



